During our minerals unit, we read a section about the physical properties of minerals. The hardness, streak, luster, specific gravity, cleavage, fracture and special properties of a mineral can be used to distinguish it from others that may have a similar appearance. As a lab to complement this lesson, students performed several tests on four "mystery mineral" samples, then had to make hypotheses about the identity of each.
To prepare for the assignment, I created mineral identification kits that included a magnifying glass, a streak plate, a piece of copper, an iron nail, a magnet, and a piece of glass. Because I grouped each class into four groups, I chose four "mystery minerals" to rotate around the room. I chose to include gypsum (for its unusual softness), sulfur (for its unusual appearance), sphalerite (for its unusual reaction to hydrochloric acid), and chalcopyrite (for its sparkly, metallic luster). Later on, we included some samples of calcite just for fun.
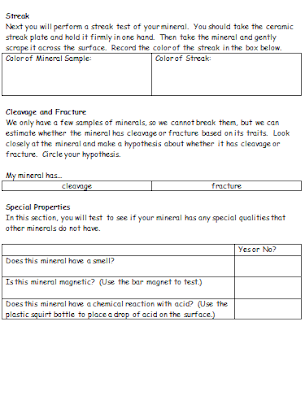
Students received a lab packet that walked them through the tests and observations that needed to be recorded. Each group made observations and sketches about the appearance, luster and texture of the minerals. They also performed a streak test on the mineral with the ceramic streak plate, as well as a hardness test with fingernails, pieces of copper, iron nails, pieces of glass and ceramic streak plates. A final section of the packet required that students look for other unique properties, such as magnetism, residue/greasiness, and chemical reactions with acid.
For obvious safety reasons, we performed the acid tests as a whole group with adults applying the hydrochloric acid to the samples. For sulfur, gypsum and chalcopyrite, there is no real reaction or effect when the acid is applied. Sphalerite, however, creates a very noticeable rotten egg odor when hydrochloric acid is applied and it will eventually dissolve if enough acid is used. At this point, we added samples of calcite to the mix because they actually fizz with just a few drops of acid.
At the end of the testing phase, I created a chart, shown below, with the officially recognized geologists' data for each mineral. Students then compared their results to the results in the chart and tried to match each sample to its true identity.
I'm not quite sure why, but this is always one of my favorite labs of the year. We may do many active lessons in this class, but this one really feels like "real science" to the kids because they are using real scientific tools and materials to find real scientific data. At this level of science, most of the so-called "labs" that we complete are really just demonstrations or models, but in this case, it just feels more inquiry-based. Best of all, it can be completed with just a few common objects and a box of mineral samples from your typical school supply catalog!
Subscribe to:
Post Comments (Atom)












0 comments:
Post a Comment